Mapping the "immunity" of human health
Vision and infrastructure
ZoonoSense is designed to be the underlying infrastructure for the next generation of precision medicine. Our vision is to map an "immunity map" of human health and achieve this through a clear three-stage path to AI-driven growth.
Phase 1:
Clinical commercialization has been implemented
Our current focus is on the commercialization of two high-value applications: early tumor screening and organ transplant monitoring. By addressing these critical clinical needs, we are building a solid revenue base and establishing a clinical presence in the medical community.
Phase 2:
Field expansion and integration of pharmaceutical companies
In the medium-term plan, we will expand our business scope to the full-cycle management of autoimmune diseases and neurological diseases. At the same time, we will establish in-depth strategic cooperation with the world's top pharmaceutical companies to directly embed ZoonoSense into their new drug development and clinical trial processes to help accelerate the development of therapies.
Stage 3:
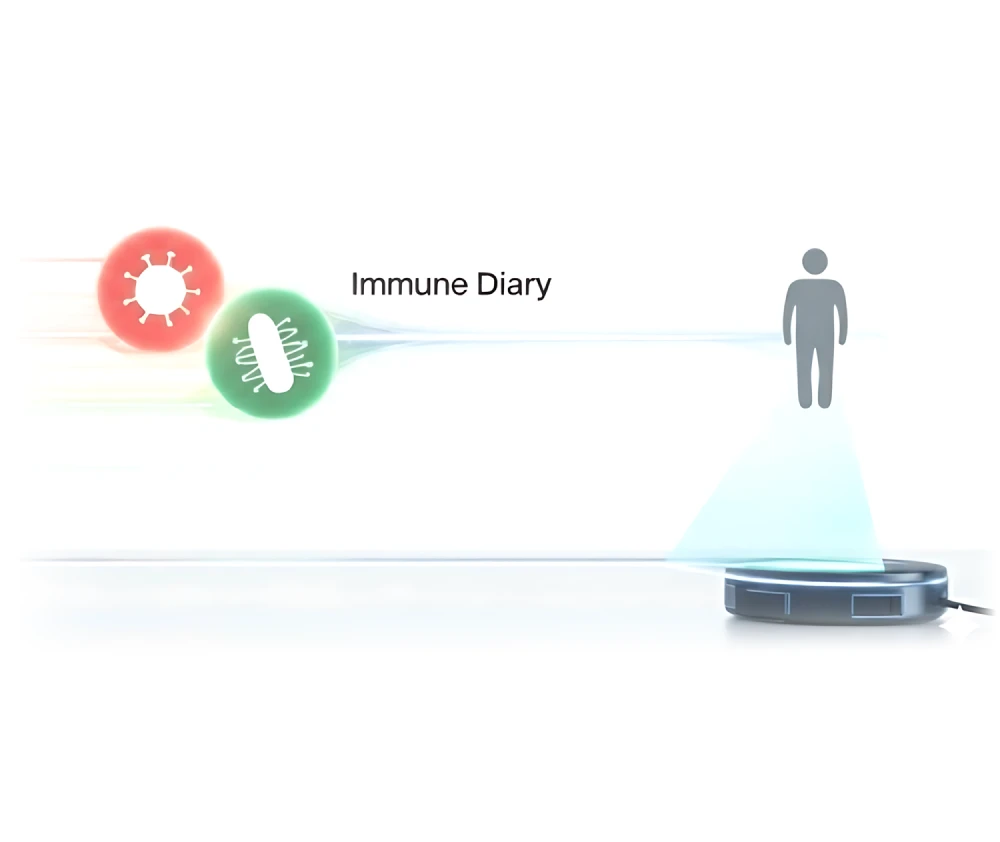
"Immunity Diary" and Paradigm Shift
Our ultimate goal is to create a dynamically updated "map of human immunity" that covers the entire life cycle. We will create a personalized "immunization diary" for each person. This represents a fundamental shift in the healthcare system from passive "treatment of diseases" to active "management of health".
ZoonoSense™ breaks through the "impossible triangle" of early cancer screening
This is the