Generative molecular design engine
Generative Molecular Design Engine
Deep integration of AI and biology
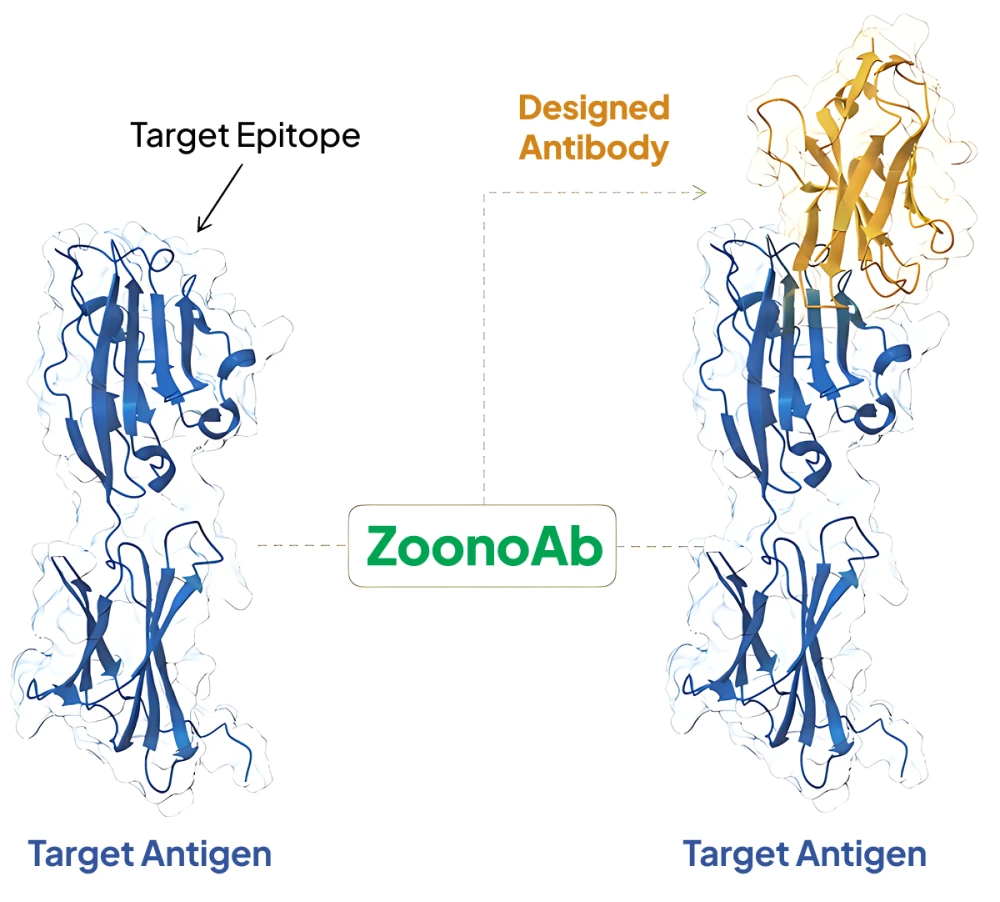
The core of our platform is ZoonoAb™, a multimodal generative model system designed to bridge the gap between computational prediction and clinical applications. ZoonoAb™ re-defines the discovery process of biologics by training on massive amounts of protein sequence and structural data, enriched with our unique, biologically significant wet experimental data.
ZoonoAb™ is built upon Zoonogen’s extensive experience in monoclonal antibody discovery for both academic and industrial applications, powered by proprietary algorithms. Our hybridoma-based monoclonal antibody database (ZMDB™) integrates biological phenotypic data with immunoglobulin (Ig) genetic information, enabling ZoonoAb™ to rapidly design highly optimized antibodies for downstream development.
01
In-Vivo Data generation
at Large-Scale
Rapid Scaling Human-Relevant Data
Our experimental infrastructure is designed to achieve a core goal: to teach ZoonoAb™ how to succeed in the first place in human biology. We produce thousands of antibody designs directly in mammalian systems and measure their binding affinity, full set of druggability parameters, and functional activity in the real cellular environment.
This tightly integrated feedback closed loop creates unique advantages. By training with data that is highly reflective of the human biological environment, we have greatly improved ZoonoAb™'s ability to design molecules with "human-ready" properties from the start, effectively eliminating the laboratory screening process of traditional methods.
02
Parallelized De Novo
Antibody design
De Novo Antibody Design with a Batch of Targets
ZoonoAb's extremely high hit rate on soluble targets have revolutionized the pace of drug discovery. We can now work on multiple parallel projects simultaneously, designing multiple targets in the time which it takes to deliver only one molecule using traditional methods.
Unlike animal immunization or phage display techniques that rely on randomness, ZoonoAb™ allows us to choose the specific binding epitopes. Each set of designs is precisely mapped to a unique pre-selected site on the target. This enable us to systematically explore the biology of the epitope and reveal functional differences between different sites, thereby guiding the formulation of downstream treatment strategies with greatly improved speed and precision.